Gold nanoparticles
Gold nanoparticles (GNPs) are a promising technology with applications in a wide range of fields including catalysis, electronics, materials science, and healthcare. They are of great interest to researchers because of their unusual optical, electronic, and chemical properties.
They can also be easily synthesized in a variety of shapes, including spheres, rods, and stars, with sizes ranging from 1 – 100 nm. They are produced as a suspension – the particles are suspended in a solvent, most often water.
Unique Optical Properties
One of the most useful optical properties of GNPs is that they change color readily, depending on their size, shape, and charge. This makes them, among other things, excellent labels for colorimetric detection of biomolecules.
These optical properties derive from an effect called surface plasmon resonance. Like all metals, gold contains free-moving electrons. When light hits the surface of a nanoparticle, these free electrons interact with the electric fields of the light rays and produce oscillations of charge that resonate with the wavelength of visible light.
The result is that GNPs absorb and reflect light at certain wavelengths, depending on their size, shape and surface chemistry.
For example, small (around 30 nm) GNPs absorb light in the blue-green portion of the spectrum (around 450 nm) and reflect red light (around 700 nm), so they are a rich red color.
As particle size increases, solutions become pale blue or purple as the red light is absorbed and blue light is reflected, until most visible wavelengths are reflected, at which point the solution is translucent.
Applications
Binding Properties
Another key property of GNPs is that they bind strongly to a range of molecules. This means that they can be coated with all kinds of molecules, such as polymers or biological recognition compounds, and their surfaces are tailored for specific applications.
For example, by coating GNPs with antibodies that bind to specific biomarkers, then measuring how the nanoparticles absorb light, the technology could be used to diagnose cancers and infectious agents.
Biomedical Applications
GNPs are ideally suited for biomedical research because they are biologically inert and generally considered to be non-toxic. There are four main areas of focus: medical imaging, diagnosis, drug delivery and targeted killing of cells.
Diagnostics
Gold is a popular choice for diagnostics because it binds strongly to short, single strands of RNA or DNA (oligonucleotides) and changes color easily. By coating GNPs with oligonucleotides, they can be used to capture and identify genetic sequences that can be linked to molecules such as bacteria. Gold nanoparticles are also common in lateral flow immunoassays, a common household example being the home pregnancy test.
Drug Delivery
Because it is easy to attach molecules to gold, the particles can act as drug delivery vehicles, carrying drugs inside tumors, for example. Nanoparticles get trapped in the porous network of blood vessels that feed a tumor and accumulate there. When light shines on them, they absorb near-infrared wavelengths that pass through tissue without causing harm and start generating heat. This heat can kill cancer cells or release drugs from carriers.
Another approach is to build scaffolds out of GNPs and then arrange DNA or RNA around them (called spherical nucleic acids, or SNAs). They pass easily through the skin’s top layer – offering the potential for treatments for melanoma and other skin conditions – and are also able to cross the blood–brain barrier, so they could be used to target brain tumor cells.
Electronics, Food Science and Other Applications
Apart from life sciences, GNPs can be used in many other areas. These include in electronics as conductors and connectors in products such as printable inks and electronic chips; and in a variety of sensors. For example, a colorimetric sensor based on gold nanoparticles can identify if foods have started to go off
Other methods, such as surface enhanced Raman spectroscopy, rely on gold nanoparticles as substrates to enable the measurement of vibrational energies of chemical bonds. This technique could be used to detect pollutants and other molecules.
Although gold in bulk is a poor catalyst, it’s a different story for GNPs, which are proving to be important catalysts in fundamental research, in green chemistry where room temperature conversion of biomass and pollutants are crucial, and for fuel cell applications.
For example, GNPs coated onto semiconductor metal oxides catalyze the oxidation of carbon monoxide at relatively low temperatures. In fact, gold catalysts can promote many reactions, at lower temperatures and with higher selectivity than other metal catalysts.
Other examples include the selective oxidation of propylene, alcohols and polyols; selective hydrogenation; and hydrochlorination.
.jpg)
Suppliers for the Life Sciences Industry
There are many companies operating globally to serve the life sciences market. These include Cytodiagnostics, Sigma-Aldrich, BBI Solutions and Nanosphere.
Sigma-Aldrich, in conjunction with Cytodiagnostics, a biotech company based in Ontario, Canada, offers a broad portfolio of gold nanoparticles geared specifically for high-tech applications within life science and materials science. GNPs are available in sizes ranging from 5 – 400 nm in diameter with numerous surface functionalities in a variety of solvent compositions.
While spherical gold nanoparticles are traditionally made using reducing agents such as sodium citrate or sodium borohydride, Cytodiagnostics has a propriety process and formulation to prepare highly spherical gold nanoparticles, without harsh reducing agents. They claim that their proprietary protocols produce particles with uniform shapes and narrow size distributions.
The UK company BBI Solutions offers a range of particle sizes from 2 – 250 nm for a range of applications. BBI says its unique manufacturing technique allows the production of large batches of gold to a high level of reproducibility of size, dispersion, and shape. It claims its gold manufacturing technique guarantees: consistency; high stability; scalability; and quality.
US company Nanosphere, now part of Luminex Corporation, has developed a detector called Verigene based on GNPs coated with oligonucleotides to identify a dozen bacteria known to cause infection. In some cases, it can do this in 2 – 3 hours. Delivery of this time-critical information enables clinicians to provide targeted patient care more quickly than waiting for the results of cultured samples. Verigene is designed to target infections in the bloodstream, respiratory tract, and gastrointestinal tract.
The Future for Life Sciences Applications
Cancer therapy continues to be a major area of interest. An array of approaches are under investigation. These include delivering cancer medication, such as tumour necrosis factor, using GNPs (AstraZeneca/CytImmune); developing SNAs to pass from the bloodstream into the brain to treat brain tumours (AuraSense Therapeutics) and other solid tumours; coating glass shells with GNPs to improve the aim of lasers used to image and zap tumours (Nanospectra Biosciences).
These nanoshells could also serve as a delivery vector for gene silencing (when a gene is switched off). They can carry specific strands of DNA oligonucleotides or RNA molecules that are released when they are exposed to ultraviolet light, and turn off expression of a gene.
Much work continues to focus on diagnostics. Examples include GNP strip tests to detect certain heart attacks by identifying cardiac troponin I (cTn-I), found at several thousand times higher in patients experiencing myocardial infarctions (New York University Polytechnic School of Engineering); flu tests consisting of GNPs coated with antibodies that bind to specific strains of the flu virus (University of Georgia); and a new device that can spot the volatile organic compounds in exhaled breaths of patients with lung cancer (University of Colorado).
References and Further Reading
Gold Nanoparticles: Properties and Applications
Biomedicine: The New Gold Standard
Gold Nanoparticles Market Size to Exceed USD 8 Billion by 2022
Graphene and its derivatives represent an exciting area of chemical discovery. With it’s range of exciting properties graphene based technology has the potential to transform our lives.
Graphene oxide membranes have been receiving attention for their extremely powerful separation abilities and the ease at which it can be modified, allowing for membrane permittivity to be fine-tuned. These membranes show the potential to be used for water purification, ‘green’ gas purification and greenhouse gas capture.
AZoNano spoke to Dr. Yang Su, of the University of Manchester’s prestigious National Graphene Institute, about the work he, and the rest of the team he is part of, are conducting on these revolutionary membranes.
Why is graphene oxide a powerful candidate for use in next-generation membranes?
Next-generation membranes need to be highly selective, with a high permeability to a select few molecules, whilst also being inexpensive and stable enough for wide use. Graphene oxide shows the potential to be strong in all of these areas.
This is because the physical structure of graphene oxide sheets lends itself easily to being used as a membrane.
.jpg)
The structure of graphene oxide (GO). GO has oxidative ‘defects’ which break the perfect strucutre of graphene. Whilst this means GO is not as conductive as graphene the holes resulting from these defects make it perfect for use in membrane technology.
There are numerous pores inside membranes. It is the shape and size of these pores that defines the selectivity and permittivity of a membrane. Whereas the identity of the materials used to construct the membrane and the way in which it is processed determines its stability and cost.
Graphene oxide (GO) membranes consist of many layers of two dimensional graphene oxide layers stacked on top of each other to give a laminate of two dimensional GO sheets. These stacked sheets have interconnected channels running through them which act as the membranes pores.
The pores are uniform in size and only 0.9 nm in width. This small width means the membrane is highly selective as only ions and molecules smaller than 0.9 nm can permeate through. Any ions or molecules larger than 0.9 nm are prevented from passing through the membrane by a process called physical extrusion.
The channels inside the GO membrane can be modified with different methods based on what type of membrane is required. Modifying the membrane channels allows membrane permeability to be fine-tuned meaning certain compounds and ions can be selected in such a way that the membrane is highly permeable to them.
Additionally, unlike polymer membranes, GO membranes are more chemically inert, which means they have a longer service lifetime.
Graphene oxide is not as expensive as pure graphene and it can, in fact, be made easily using only graphite powder and some inexpensive chemistry which could even be carried out at home. (though I wouldn’t recommend it!)
This high selectivity coupled with a low cost and long operational lifetime is why there is so much interest in GO advanced membrane technology.
.jpg)
An image of multilayer graphene. Sheets in GO membranes are organised in a similar fashion. Layered one over another to give a laminate. Shutterstock | Bessarab
Can unadulterated, pure graphene be used for this purpose?
Pure graphene is highly impermeable. Graphene will occasionally be permeable to very small particles such as protons (one of the components of an atomic nucleus) but this is only under very specific conditions that you wouldn’t find outside of a lab.
Any matter larger than a proton, such as liquids and gases, will be completely impermeable to graphene. However, this impermeability has its own uses – for example graphene would work very well as a barrier or protective membrane.
.jpg)
As pure graphene is completely defect free there are no gaps in its structure making it completely impervious to all but the smallest of particles. Shutterstock | ktsdesign
What molecules and liquids are GO membranes capable of separating?
The unique pore structure of GO membrane provides a good platform for us to play with. By changing the structure of the membrane we can adjust the membranes permeability with respect to different molecules.
Depending on the membrane structure they can be used for gas separation, such as hydrogen purification, CO2 capture, or gas drying by removing water.
The GO membranes can also be used for the separation of aqueous mixtures, such as in the dehydration and purification of organic solvents. This technology has the potential to be used to separate certain solute/ions from solvents, which could potentially be used for water purification.
How can the membrane separation properties be modified? Can the membrane be functionalised or the pore size controlled in other ways?
The membrane separation properties can be modified using many different methods with the main methods being chemical functionalization and physical pore size control.
Attaching external molecules to the sheets by chemical functionalization can be used to expand the pore sizes. This results in faster transport through the membranes and different selectivities.
In contrast, our recent results show that by removing oxygen atoms from GO sheets we could induce the channels to collapse. This type of GO membrane could be used as a barrier or protective coating for the packaging industry.
.jpg)
How do you see this technology impacting the real world?
Due to its amazing separation properties we expect this technology to have a huge impact in the fields of energy reduction and environmental conservation.
For example, this technology could be used for the dehydration and purification of biofuels. Currently water is produced as a side product when biofuels are produced. This water damages the biofuel yield and the final product quality, and its removal is difficult. Our GO membranes could help solve this problem.
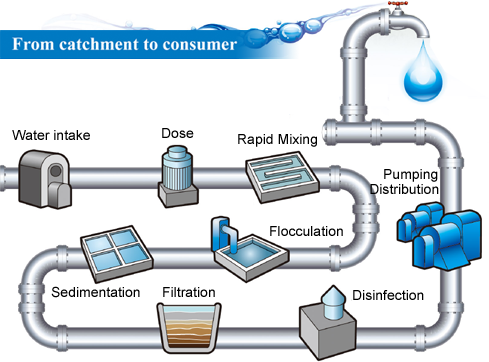
GO membranes also have the potential to be used for portable water purifiers. The small pore size of the membranes means all of the bacteria present in dirty water, and most of the other impurities, will be sieved out with only pure water passing through. This technology would be hugely useful in the developing world. The membranes also show promise for use in water desalination, but we’re not quite there yet.
The membranes also have the ability to separate gases, meaning in the future they could be used to control greenhouse gas emissions and to purify hydrogen related clean energy gases.
.jpg)
GO membrane technology could assist in the production and purification of clean biofuels.
Do you think graphene oxide membranes have the potential to be used for large scale water filtration or do you believe this would be impractical?
The membrane technology that we are developing could definitely be used for large scale water filtration.
Firstly, graphene oxide can be manufactured on a large scale meaning the materials are readily available. Secondly, the membranes are produced with little difficulty, even simple membranes formed by evaporating water from a GO suspension results in a GO membrane that performs well.
Finally, and most importantly, the membranes already show amazing properties for sieving impurities from water. Of course, at our current stage, we still need to work hard to address several issues on its practical applications, but our research team are working on this and I’m confident we will get there.
How does your research on graphene membranes fit into the wider picture, with the other research being undertaken at the National Graphene Institute?
.jpg)
The National Graphene Institute at the University of Manchester
When do you expect this technology to become widely available? What obstacles must be overcome before this happens?
We don’t have a detailed timeline yet. At our current stage, among a lot of possible applications, we are assessing them for one or two applications which could be realized on a large scale.
Our main obstacle is that we need more industrial partners to fill the gap between the lab experiments and pilot scale productions, we then could conduct application-oriented research to finalize the end products.